by Michael J. Mottl
Dept. of Oceanography, University of Hawaii, 1000 Pope Road, Honolulu HI 96822 USA
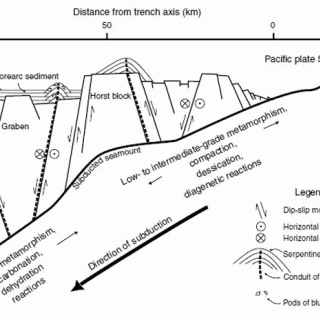
Probably the highest pH ever measured in the deep sea, and possibly in any natural setting, is that of pore water ascending through the serpentinite mud volcanoes in the Mariana forearc (Figure 1). These mud volcanoes were discovered by my colleague Dr. Patty Fryer in the mid-1970's, while completing field work with Don Hussong, then at the University of Hawaii, as part of her doctoral thesis. It was a great surprise to find what appeared to be volcanoes there, as these were supposed to belong properly in the arc, and not in the forearc. Further investigation over the past 30 years (Fryer and Salisbury, 2006) has demonstrated that these are mud volcanoes, similar structurally to conventional volcanoes except that they are cold, and are formed and fed by a rising column of serpentinized peridotite rather than by molten rock. The serpentinite is mobilized from the lithospheric mantle of the overriding Philippine plate, by water and other volatiles sweated off the westward subducting Pacific plate as it is pressurized and heated on its descent (Fryer et al., 1999; Figure 2). Because this mantle has been melted to yield crust apparently multiple times in its history, it is highly depleted in crustal components, so that the source rock of the serpentinite is depleted harzburgite. This is a rock comprised mainly of olivine and orthopyroxene, and is about as chemically simple an igneous rock as can be found on or in the seafloor.
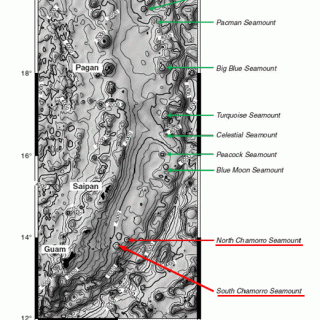
The mud volcanoes produced by this process are big: the largest, Big Blue seamount, is 2 km high and 50 km across. There are dozens of these mud volcanoes in the Mariana forearc, all within the outer half, along a 600 km-swath from about 50 to 90 km behind the trench (e.g. Figure 3). They form when the low-density serpentinite rises buoyantly along intersecting fractures and extrudes at the seafloor, though a central conduit that is narrow relative to the diameter of the volcano. This conduit feeds flows of unconsolidated sedimentary serpentinite that cascade from the summit down the slopes of the building volcano to distances of 18 km. These flows comprise the bulk of the seamount and contain variably serpentinized clasts of harzburgite ranging in size from silt to boulders. The upwelling serpentinite brings up fragments of subducted ocean crust metamorphosed in the blueschist facies, the first ever found in a modern tectonic setting (Maekawa et al., 1993).
Beginning in 1989, when we first drilled one of these mud volcanoes on Ocean Drilling Program (ODP) Leg 125, we have sampled more than a dozen of them at depth below the seafloor, by drilling (again on ODP Leg 195), piston and gravity coring, and push coring from manned submersibles (both Alvin and Shinkai-6500) and from the ROV Jason. These precision tools allowed us to sample both at depth and at seafloor springs, for the waters responsible for serpentinization, derived by dehydration of the subducting Pacific plate, ascend through the rising column of serpentinite mud that feeds the volcano and exit as cold springs on its summit. These springs, in turn, form chimneys of chemical precipitate and support both chemosynthetic microfauna and macrofaunal assemblages at the springs (Mottl et al., 2003), analogous to those found at hot springs along the mid-ocean ridge axis.
The ascending waters, as sampled both within the seafloor and at the springs, are among the most unusual waters ever sampled in the deep sea. Because of the simplicity of their depleted-harzburgite matrix, the upwelling pore waters retain a clear chemical signal of their deep metamorphic origin (Mottl, 1992) in spite of their long ascent, which ranges from 15 to 28 km from the top of the subducting plate to the seafloor. Because the waters originate by dehydration of the subducting slab, they are nearly always fresher (chlorinity of 235 to 546 mmol/kg) than the ambient bottom seawater (542 mmol/kg). Their chemistry varies abruptly with distance across the forearc (Mottl et al., 2004), and hence with depth to the top of the subducting plate: near the trench, at 48-54 km, they have pH 10.7, much higher Ca and Sr than seawater and much lower alkalinity, sulfate, Na/Cl, K, Rb, and B. These proximal springs form chimneys of brucite, Mg(OH)2. Farther from the trench, at 70 to 90 km, the waters have pH 12.5 and show the opposite trends relative to seawater for all of these species. These distal springs form chimneys and crusts of CaCO3. Seamounts at intermediate distances from the trench have intermediate values. Sulfate, Na/Cl, K, Rb, Cs, and B all increase regularly with distance from the trench, leached from the subducting sediment and altered basalt in response to increasing temperature at depth from ~100-350°C.
The pH, alkalinity, and methane content of the springs increase abruptly with distance between 66 and 71 km from the trench, corresponding to depths of 21 to 22 km to the top of the subducting plate. As methane climbs from 44 mmol/kg, and alkalinity from 3 to 69 meq/kg, pH increases abruptly from 10.7 to 12.5, as measured at 25°C. We have measured pH in excess of 12 at three mud volcanoes farthest from the trench, in seven different ODP holes at two sites (780 and 1200), in one gravity core, one piston core, and one Jason push core, for a total of 54 pore water samples. Of these, 38 are in excess of 12.3. The deepest samples, most representative of the upwelling end-member water, have pH as high as 12.60.
How do these waters achieve such high pH? While it is well known that serpentinization produces high-pH waters, these values are exceptionally high even for this setting. Solutions involved in serpentinization evolve toward high pH primarily because peridotites contain so little Al and have a low ratio of Si to Mg. Weathering of silicate rocks in general consumes H+, causing solutions to become more alkaline. The pH of the weathering solution rises until it becomes saturated with secondary aluminosilicates such as clay minerals, which buffer the pH at near neutrality. When Al and Si are scarce aluminosilicates do not form, and pH rises with dissolution of olivine and orthopyroxene until brucite saturation is reached. As modeled by Palandri and Reed (2004), this occurs at a pH of 10.3 at 25ºC, in the simple system MgO-SiO2-H2O-CO2, a value close to the 10.7 we measured in pore water ascending beneath the mud volcanoes closer to the trench. If CaO is added pH can rise as high as 12.0, but if FeO and Fe2O3 are added the maximum pH at 25ºC drops again to 11.6. This compares with a range of values in natural waters from serpentinites of 10.7-11.8 (Palandri and Reed, 2004). The highest pH25ºC measured at the ultramafic-hosted hydrothermal system at Lost City on the Mid-Atlantic Ridge is 11 (Kelley et al., 2005).
We have consistently measured pH25ºC in the Mariana forearc pore waters well in excess of these values at the three distal seamounts, 70-90 km from the trench: Big Blue (pH 12.34; 9 samples >12.0), South Chamorro (pH 12.49; 39 samples >12.0), and Conical (pH 12.60; 6 samples >12.0). Springs at these distal seamounts have low Ca, in contrast to the high Ca at the proximal sites where pH is lower; this is exactly opposite to the prediction of Palandri and Reed (2004) that high Ca promotes high pH. The pH25ºC of 12.5 measured in the deep upwelling water at South Chamorro Seamount on ODP Leg 195 becomes 13.1 ± 0.1 at the in-situ temperature of 2.4ºC, calculated using PHREEQC (Parkhurst and Appelo, 1999)(and the pH25ºC of 10.7 measured at springs proximal to the trench becomes 11.1). These are the highest pH values ever measured in the deep sea, or in any other natural environment that I know of. They are equivalent to that maintained by a Ca(OH)2 buffer. Whereas the pH of 10.7 at the proximal springs can readily be explained by serpentinization, the much higher pH of the distal springs clearly requires an additional explanation.
We hypothesize that the abrupt increases in pH, alkalinity, and methane that occur between 66 and 71 km from the trench have a common cause, namely, dissolution of carbonate minerals in the subducting plate (Mottl et al., 2008). Carbonate dissolution apparently commences at 21-22 km below the seafloor at an estimated temperature of 200-250°C, and joins dehydration as a major process that devolatilizes the subducting plate. Whereas CaCO3 minerals become less soluble with increasing temperature at low pressure, their solubility increases with temperature at pressures of 3 kbar and higher (Caciagli and Manning, 2003), equivalent to burial depths of 10 km and more. Dissolution of carbonate minerals increases the alkalinity of the ascending solutions.
Besides generating high pH, serpentinization is well known to generate large quantities of H2 by oxidation of Fe2+ in olivine and orthopyroxene to Fe3+ in magnetite. This H2 will react with the dissolved carbonate now added to the ascending alkaline solutions and reduce it to methane via the reaction:
4H2 + CO32- = CH4 + H2O + 2OH- |
Note that total alkalinity, enhanced by carbonate dissolution, is conserved in this reaction, but that the original carbonate alkalinity is replaced by hydroxyl alkalinity, which increases pH. At the same time the carbonate-C is reduced to methane. This reaction neatly accounts stoichiometrically for the much higher pH of the distal springs and the fact that most of the ascending C is as methane rather than dissolved carbonate.
But why is the transition to carbonate dissolution so abrupt? The transition occurs ~70 km from the trench, where metabasites recovered from the serpentinite mud indicate the transition from lawsonite-blueschist to epidote-blueschist facies also occurs. Thermodynamic modeling indicates that replacement of lawsonite by epidote, which occurs at a temperature of ~223ºC for the chemically simple end-member epidote clinozoisite, causes an abrupt increase in the Na/Ca ratio in solution. As the major cation in solution Na cannot vary much, as its concentration is largely fixed by charge balance. The abrupt increase in Na/Ca is thus accomplished mainly by a drastic decrease in Ca2+: the epidote effectively sucks most of the Ca out of solution. Because the solutions are saturated with CaCO3, and appear to remain so throughout their long ascent, the drop in Ca2+ is accompanied by a large increase in carbonate ion, which can only be supplied by massive dissolution of CaCO3 at the top of the subducting slab. We hypothesize, therefore, that the abrupt increase in dissolution of CaCO3 is triggered by the transition with increasing depth from the lawsonite- to the epidote-blueschist facies, which in turn triggers the transition from a high pH25ºC of 10.7 to an extremely high pH25ºC of 12.5.
References
Caciagli, N.C., and C.E. Manning (2003) 'The solubility of calcite in water at 6-16 kbar and 500-800ºC. Contrib. Mineral. Petrol. 146:275-285.
Fryer, P.B., and M.H. Salisbury (2006) Leg 195 Synthesis: Site 1200--Serpentitine Seamounts of the Izu-Bonin/Mariana Convergent Plate Margin, ODP Leg 125 and 195 Drilling Results. Proc. Ocean Drill. Program Sci. Results 195:1-30.
Fryer, P., C.G. Wheat, and M.J. Mottl (1999) Mariana blueschist mud volcanism: implications for conditions within the subduction zone. Geology 27:103-106.
Maekawa, H., M. Shozui, T. Ishii, P. Fryer, and J.A. Pearce (1993) Blueschist metamorphism in an active subduction zone. Nature 364:520-523.
Mottl, M.J. (1992) Pore waters from serpentinite seamounts in the Mariana and Izu-Bonin forearcs, Leg 125: evidence for volatiles from the subducting slab. Proc. Ocean Drill. Program Sci. Results 125:373-385.
Mottl, M.J., S.C. Komor, P. Fryer, and C.L. Moyer (2003) Deep-slab fluids fuel extremophilic Archaea on a Mariana forearc serpentinite mud volcano: Ocean Drilling Program Leg 195. Geochemistry, Geophysics, Geosystems 4(11):9009, doi:10.1029/2003GC000588, 14pp.
Mottl, M.J., C.G. Wheat, P. Fryer, J.Gharib, and J.B. Martin (2004) Chemistry of springs across the Mariana forearc shows progressive devolatilization of the subducting plate. Geochim. Cosmochim. Acta 68:4915-4933.
Mottl, M.J., T.M. McCollom, C.G. Wheat, and P. Fryer (2008) Decarbonation of the Subducting Pacific Plate Triggered by the Lawsonite-to-Epidote Transition Beneath the Mariana Forearc Serpentinite Mud Volcanoes (abstract). American Geophysical Union Fall Meeting, U51C-03.
Salisbury, M.H., M. Shinohara, C. Richter, et al., (2002) Proceedings of the Ocean Drilling Progarm, Initial Reports Volume 195.
Figure Captions
Figure 1. Bathymetry of the southern Mariana arc and locations of selected serpentinite mud volcanoes (Salisbury et al., 2002). Figure 3 shows a detailed side-scan sonar image of the North and South Chammo Seamounts. The term 'seamount' is descriptive whereas 'mud volcano' is interpretive.
Figure 2. A conceptual sketch of how serpentinite mud volcanoes form in the forearc of the Marianas subduction complex. Fluids from heated and pressurized sediments on the underthrust plate flow up through faulted harzburgite in the overthrust mantle wedge. Slurries of serpentinized harzburgite erupt on the seafloor to form and enlarge serpentinite mud volcanoes. (Salisbury et al., 2002).
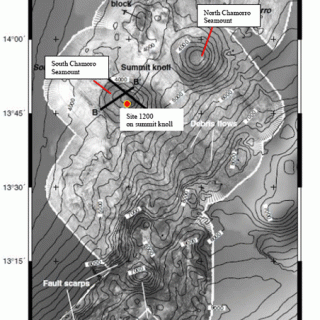
Figure 3. Sidescan sonar image of the North and South Chamorro seamounts. The water depth is about 2920 meters. Rock cores and pore fluids were sampled at depths down to 50.4m beneath the seafloor at Site 1200 on the South Chamorro seamount knoll. This work was accomplished on Leg 195 of the Ocean Drilling Program (Salisbury et al., 2002).